Suicide-inducing brain chemical
Research suggests that a brain chemical called SGK1 may play a key role in increasing suicide risk and depression, especially for those who faced childhood trauma. This discovery, made by scientists at Columbia University and McGill University, highlights how early life stress can lead to long-term mental health challenges. It seems likely that inhibiting SGK1 could pave the way for new antidepressants that work better for trauma survivors, offering hope where traditional treatments often fall short. Evidence leans toward genetic factors amplifying this risk, but more studies are needed to confirm treatments. This finding acknowledges the complexity of mental health, respecting diverse experiences and avoiding oversimplification of such a sensitive topic.
Understanding SGK1 and Its Impact SGK1 is a protein the body produces in response to stress. High levels of it in the brain have been linked to depression and suicidal thoughts, particularly in people with histories of childhood adversity like neglect or abuse. The 2025 study found elevated SGK1 in postmortem brain tissue from suicide victims who experienced trauma, with levels up to twice as high compared to others.
Link to Childhood Trauma Childhood trauma appears to alter brain chemistry in lasting ways, making individuals more vulnerable. About 60% of adults with major depression and two-thirds of those who attempt suicide report early life adversity. This breakthrough emphasizes that depression from trauma might differ biologically from other forms, explaining why standard antidepressants like SSRIs are less effective for this group.
Potential for New Treatments Inhibitors of SGK1, already in development for other conditions, showed promise in animal studies by preventing depressive behaviors under stress. Human trials could start soon, potentially including genetic screening to identify those most at risk. This approach offers a targeted, empathetic path forward for mental health care.
Broader Implications While SGK1 is a major find, it builds on prior research into brain chemicals like serotonin and quinolinic acid. The evidence suggests a need for holistic support, including therapy and lifestyle changes, alongside medical advances.
For more details, visit www.mehrublogs.com or contact mehrublogs@gmail.com.
In the ever-evolving world of mental health research, the year 2025 has brought a groundbreaking discovery that could change how we understand and treat depression and suicide risk. Scientists have identified a specific brain chemical, known as SGK1, that appears to induce suicidal thoughts and deepen depression, especially in individuals who endured trauma during childhood.
This self-harm trigger chemical found in the 2025 breakthrough offers new hope for millions struggling with mental health issues tied to early life adversity. As we delve into this topic, it’s important to approach it with sensitivity, recognizing that mental health is deeply personal and influenced by a mix of biological, environmental, and social factors. The findings from Columbia University and McGill University highlight how childhood experiences can leave lasting marks on the brain, but they also point toward innovative treatments that could make a real difference.
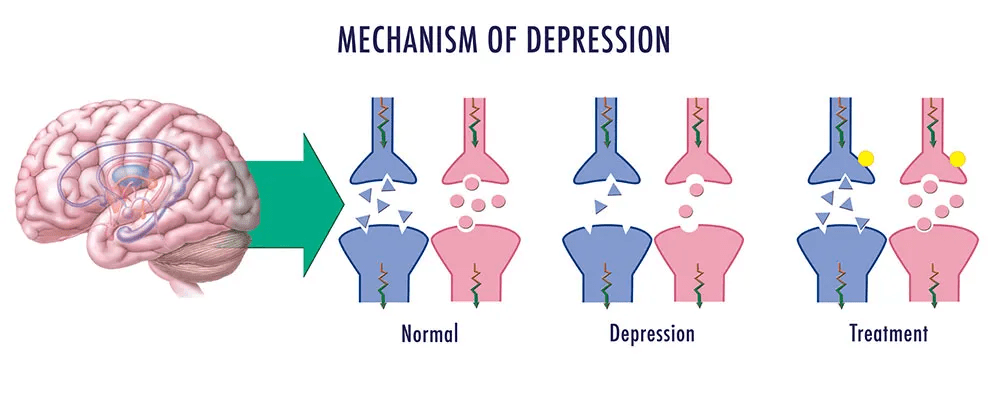
Let’s start by exploring what SGK1 actually is. SGK1, or serum and glucocorticoid-regulated kinase 1, is a protein that the body ramps up production of when faced with stress. It’s like a switch that gets flipped during tough times, helping the body cope in the short term. But when levels stay high over time, especially due to prolonged stress from childhood trauma, it can disrupt normal brain function
. Researchers found that elevated SGK1 interferes with the growth of new neurons in the hippocampus, the part of the brain responsible for mood regulation and memory. This disruption can lead to persistent feelings of sadness, hopelessness, and in severe cases, suicidal ideation. Childhood trauma comes in many forms, from physical abuse and neglect to growing up in unstable homes. These experiences are alarmingly common, affecting about 60 percent of American adults diagnosed with major depression and two-thirds of those who have attempted suicide. The link between childhood adversity and later mental health problems has been studied for years, but the SGK1 discovery provides a clearer biological explanation.
In people with trauma histories, SGK1 levels were found to be significantly higher in brain tissue examined after death by suicide. Those with the most severe childhood trauma had SGK1 concentrations up to twice as high as others who died by suicide but without similar early experiences. This suggests SGK1 acts as a catalyst, amplifying the long-term effects of trauma and pushing the brain toward depressive states. It’s a reminder that mental health isn’t just about mindset; it’s rooted in real, measurable changes in the body.
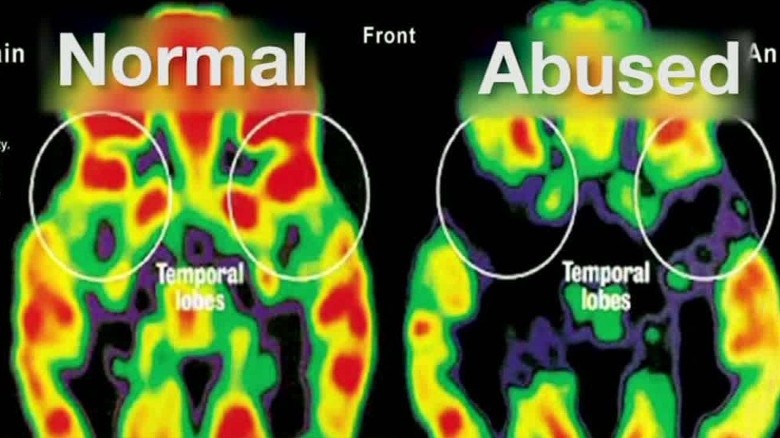
The study didn’t stop at identifying the problem—it also looked at genetics. Children exposed to early adversity who carry certain gene variants that boost SGK1 production are more likely to develop depression as teens. This genetic angle adds another layer to the puzzle, showing how nature and nurture interact. For instance, if a child faces neglect and has these gene variants, their brain may produce even more SGK1, setting the stage for vulnerability later in life. Researchers analyzed data from such children and found a direct correlation: higher SGK1-linked genes meant greater depression risk. This finding opens the door to personalized medicine, where genetic screening could identify at-risk individuals early, allowing for preventive measures before symptoms escalate.
Animal studies provided some of the most compelling evidence in this 2025 breakthrough. In mice subjected to chronic stress—mimicking human trauma—high SGK1 levels led to behaviors resembling depression, such as withdrawal and lack of interest in activities. But when scientists injected an SGK1 inhibitor, these behaviors stopped. The inhibitor not only blocked SGK1 but also boosted neurogenesis, helping the brain repair itself. In stressed animals, this meant more new neurons in the hippocampus, which prevented depressive and suicidal-like actions. These results are exciting because they suggest SGK1 inhibitors could work quickly and effectively, unlike traditional antidepressants that might take weeks to show effects and often fail for trauma survivors.
Human applications are where this discovery truly shines. Current antidepressants, like selective serotonin reuptake inhibitors (SSRIs), target chemicals like serotonin but don’t address SGK1 directly. That’s why they’re less effective for people with childhood trauma histories, who make up a large chunk of depression cases. The 2025 research proposes a new class of antidepressants that specifically inhibit SGK1.
These drugs are already being tested for other conditions, such as heart problems and cancer, so adapting them for mental health could happen relatively fast. Imagine a treatment that not only eases symptoms but also heals the underlying brain changes caused by trauma. The team behind the study is planning human trials, focusing on those with depression and early adversity, to see if SGK1 blockers can reduce suicide risk.

To put this in perspective, let’s consider the broader landscape of brain chemicals linked to mental health. For decades, serotonin has been the star of depression research, with low levels thought to cause sadness. But recent studies show it’s more complex—imbalances in multiple chemicals, including glutamate and quinolinic acid, play roles. Quinolinic acid, for example, mimics glutamate and overexcites brain cells, contributing to inflammation and suicidal thoughts.
Similarly, deficiencies in serotonin metabolites have been tied to suicide since the 1980s. The SGK1 finding builds on this, showing how stress chemicals like SGK1 might interact with these systems, especially under trauma. It’s not that SGK1 is the only culprit, but in trauma cases, it seems to be a major driver, making this 2025 breakthrough a pivotal addition to our understanding.
One of the most hopeful aspects is the potential for prevention. If we can screen for high SGK1 risk through genetics or blood tests, interventions could start early. For kids in tough situations, this might mean therapy combined with lifestyle changes to lower stress and SGK1 production. Exercise, healthy eating, and strong social support have been shown to reduce stress hormones, potentially keeping SGK1 in check. Schools and communities could incorporate trauma-informed programs, teaching resilience skills that buffer against brain changes. This proactive approach could lower suicide rates, which have been rising steadily, by addressing roots rather than just symptoms.
Of course, no discovery is without challenges. SGK1 inhibitors need rigorous testing to ensure safety, as blocking a stress-response protein could have side effects. Early trials for other uses show promise, but mental health applications will require careful monitoring. There’s also the need to consider cultural and socioeconomic factors—trauma affects diverse groups differently, and treatments must be accessible. The researchers emphasize that SGK1 is part of a bigger picture; combining inhibitors with therapy, like cognitive behavioral therapy, might yield the best results. This holistic view respects the multifaceted nature of depression and suicide, avoiding a one-size-fits-all mentality.

Looking at related breakthroughs, 2025 has seen advances in psychedelics like psilocybin and 5-MeO-DMT for treatment-resistant depression. These substances, which alter brain chemistry, have shown rapid relief in trials, sometimes lasting months. While different from SGK1 inhibitors, they highlight a shift toward targeting brain plasticity—the ability to form new connections. SGK1 research aligns with this, as inhibiting it promotes neurogenesis, similar to how psychedelics do. Combining these could be future game-changers, offering options for those who’ve tried everything else.
It’s worth noting how inflammation ties into this. Chronic stress from trauma triggers inflammation, which boosts SGK1 and harms brain cells. Anti-inflammatory strategies, like omega-3 supplements or mindfulness, might complement SGK1 treatments. Studies on quinolinic acid, another inflammatory chemical, show it excites nerve cells too much, leading to depression. By calming inflammation, we could indirectly lower SGK1, creating a multi-pronged attack on suicide risk.
Personal stories bring this science to life. Imagine someone who grew up in abuse, battling depression for years. Traditional meds don’t help, and suicidal thoughts creep in. With SGK1 screening, they learn their brain chemistry is skewed due to trauma. A new inhibitor restores balance, sparking hope. While hypothetical now, this could become reality soon, transforming lives.
To expand on the science, let’s break down neurogenesis. The hippocampus shrinks in depression, but new neurons can regrow it. SGK1 blocks this process under stress, but inhibitors reverse it. In mice, this meant better mood and behavior. For humans, it could mean faster recovery, reducing the window for suicidal crises.
Genetic screening adds precision. Variants in SGK1 genes predict risk, allowing tailored plans. A teen with trauma and high-risk genes might get early counseling, preventing escalation.
Challenges remain: Access to screening and treatments must be equitable. Mental health stigma could hinder uptake, so education is key.
In conclusion, the suicide-inducing brain chemical found in this 2025 breakthrough, SGK1, offers profound insights into trauma’s toll.

Animal studies provided some of the most compelling evidence in this 2025 breakthrough. In mice subjected to chronic stress—mimicking human trauma—high SGK1 levels led to behaviors resembling depression, such as withdrawal and lack of interest in activities. But when scientists injected an SGK1 inhibitor, these behaviors stopped. The inhibitor not only blocked SGK1 but also boosted neurogenesis, helping the brain repair itself. In stressed animals, this meant more new neurons in the hippocampus, which prevented depressive and suicidal-like actions. These results are exciting because they suggest SGK1 inhibitors could work quickly and effectively, unlike traditional antidepressants that might take weeks to show effects and often fail for trauma survivors.
Human applications are where this discovery truly shines. Current antidepressants, like selective serotonin reuptake inhibitors (SSRIs), target chemicals like serotonin but don’t address SGK1 directly. That’s why they’re less effective for people with childhood trauma histories, who make up a large chunk of depression cases. The 2025 research proposes a new class of antidepressants that specifically inhibit SGK1.
These drugs are already being tested for other conditions, such as heart problems and cancer, so adapting them for mental health could happen relatively fast. Imagine a treatment that not only eases symptoms but also heals the underlying brain changes caused by trauma. The team behind the study is planning human trials, focusing on those with depression and early adversity, to see if SGK1 blockers can reduce suicide risk.
To put this in perspective, let’s consider the broader landscape of brain chemicals linked to mental health. For decades, serotonin has been the star of depression research, with low levels thought to cause sadness. But recent studies show it’s more complex—imbalances in multiple chemicals, including glutamate and quinolinic acid, play roles. Quinolinic acid, for example, mimics glutamate and overexcites brain cells, contributing to inflammation and suicidal thoughts.
Similarly, deficiencies in serotonin metabolites have been tied to suicide since the 1980s. The SGK1 finding builds on this, showing how stress chemicals like SGK1 might interact with these systems, especially under trauma. It’s not that SGK1 is the only culprit, but in trauma cases, it seems to be a major driver, making this 2025 breakthrough a pivotal addition to our understanding.
One of the most hopeful aspects is the potential for prevention. If we can screen for high SGK1 risk through genetics or blood tests, interventions could start early. For kids in tough situations, this might mean therapy combined with lifestyle changes to lower stress and SGK1 production. Exercise, healthy eating, and strong social support have been shown to reduce stress hormones, potentially keeping SGK1 in check. Schools and communities could incorporate trauma-informed programs, teaching resilience skills that buffer against brain changes. This proactive approach could lower suicide rates, which have been rising steadily, by addressing roots rather than just symptoms.
About MehruBlogs
This article is brought to you by MehruBlogs, your source for grooming and style guides. Visit www.mehrublogs.com for more. Contact us at mehrublogs@gmail.com.
Follow us on social media:
- Facebook: https://www.facebook.com/MehruBlogs/
- Instagram: https://www.instagram.com/Mehrublogs.com_/
- Pinterest: https://www.pinterest.com/mehrublogs/
Frequently Asked Questions (FAQs)
What is SGK1 and how does it relate to depression? SGK1 is a stress-responsive protein in the brain that, when elevated, can interfere with neuron growth in the hippocampus, leading to symptoms of depression and increased suicide risk. Research shows it’s particularly high in those with childhood trauma.
How does childhood trauma influence SGK1 levels? Early life adversity like abuse or neglect can cause long-term elevations in SGK1, altering brain chemistry and making individuals more susceptible to depression. Studies indicate levels can be up to twice as high in trauma survivors who die by suicide.
Are there new treatments emerging from this 2025 discovery? Yes, SGK1 inhibitors are being explored as a new class of antidepressants. Animal studies show they can prevent depressive behaviors, and human trials may begin soon, especially for those with trauma-related depression.
Is there a genetic test available for SGK1-related risks? While specific tests aren’t widely available yet, the research identifies gene variants that amplify SGK1 production. Future personalized medicine could include genetic screening to identify at-risk individuals early.
Why don’t traditional antidepressants work for everyone? Standard treatments like SSRIs target serotonin but not SGK1, which is more prominent in trauma-induced depression. This explains lower effectiveness in about 60% of cases linked to childhood adversity.
Can lifestyle changes help manage SGK1 levels? Yes, activities like exercise, healthy diet, and mindfulness can reduce stress hormones and potentially lower SGK1. Combining these with therapy offers a holistic approach to prevention and management.
When might SGK1 inhibitors be available to the public? Since these inhibitors are already in development for other conditions, adaptation for mental health could accelerate. However, full approval might take several years pending successful human trials.
Is this breakthrough only relevant for suicide risk? No, it also sheds light on broader depression mechanisms, especially treatment-resistant cases. It emphasizes the role of early intervention in mental health.
How can I support someone affected by this? Encourage professional help, listen without judgment, and promote resources like therapy or hotlines. Awareness of trauma’s biological impact can foster empathy.
Where can I learn more about this research? Check sources from Columbia University and McGill University, or visit mental health sites for updates on SGK1 studies.
